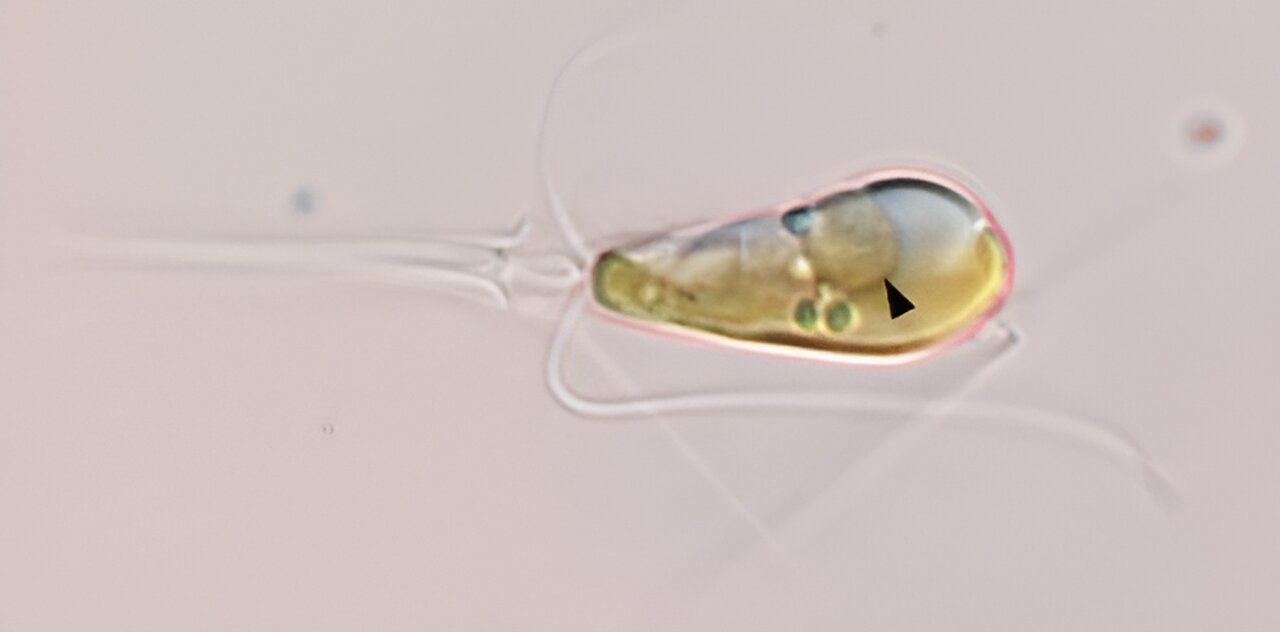
Optical micrograph shows the marine haptophyte alga Braarudosphaera bigelowii with a black arrow indicating the nitroplast organelle. Credit: Tyler Cole
× Close
Optical micrograph shows the marine haptophyte alga Braarudosphaera bigelowii with a black arrow indicating the nitroplast organelle. Credit: Tyler Cole
Modern biology textbooks assert that only bacteria are capable of taking nitrogen from the atmosphere and converting it into a habitable form. Plants that fix nitrogen, such as legumes, do so by harboring symbiotic bacteria in their root nodules. But the latest discovery turns this rule upside down.
In two recent papers, an international team of scientists describes the first known nitrogen-fixing organelle inside a eukaryotic cell. The organelle is the fourth example in the history of primary endosymbiosis—the process by which a prokaryotic cell is engulfed by a eukaryotic cell and evolves beyond the symbiosis into an organelle.
“It's very rare for organelles to arise from this kind of thing,” said Tyler Cole, a postdoctoral researcher at UC Santa Cruz and first author of one of two recent papers. “The first time we thought this happened, it gave rise to all complex life forms. Everything more complex than a bacterial cell owes its existence to this event,” he said, referring to the origins of mitochondria. “A billion years ago or so, that happened again with chloroplasts, and that gave us plants,” Cole said.
A third well-known example involves a chloroplast-like microbe. The latest discovery is the first example of a nitrogen-fixing organelle, which researchers call a nitroplast.
A decades-old mystery
Discovering the organelle took a bit of luck and decades of work. In 1998, Jonathan Zahr, a distinguished professor of marine science at the University of California, Santa Cruz, found a short DNA sequence from what appeared to be from an unknown nitrogen-fixing cyanobacteria in Pacific seawater. Zahr and his colleagues spent years studying the mysterious object, which they named UCYN-A.
Meanwhile, Kyoko Hagino, a paleontologist at Kochi University in Japan, was actively trying to culture seaweed. It turned out to be the host organism of UCYN-A. It took more than 300 sampling expeditions and more than a decade, but Hagino eventually succeeded in growing the algae in culture, allowing other researchers to begin studying UCYN-A and its marine algae host together in the laboratory.
For many years, scientists considered UCYN-A to be an endosymbiont closely related to algae. But the two recent papers suggest that UCYN-A has co-evolved with its former symbiotic host, and now fits the criteria for an organelle.
Organic origins
In a paper published in cell In March 2024, Zahr and colleagues from MIT, the Instituto de Ciencia de Barcelona, and the University of Rhode Island showed that the volume ratio between UCYN-A and their algal hosts is similar across different species of marine euphyte algae. Prarodosphaera bigeloi.
The researchers use a model to demonstrate that host cell growth and UCYN-A are controlled by the exchange of nutrients. Their metabolic processes are related. This synchronization in growth rates led researchers to call UCYN-A “organelle-like.”
“This is exactly what happens with organelles,” Zahr said. “If you look at mitochondria and chloroplasts, they are the same thing: they expand with the cell.”
A soft Credit: Valentina Loconte
× Close
A soft Credit: Valentina Loconte
But scientists did not confidently call UCYN-A an organelle until they confirmed other evidence. In the Cover article From the magazine Sciences, published today, features Zahr, Qualley, Kendra Turk Kubo, Wing-Kwan Esther Mak of the University of California, Santa Cruz, and collaborators from the University of California, San Francisco, Lawrence Berkeley National Laboratory, National Taiwan Ocean University, and Kochi University in Japan. UCYN-A imports proteins from its host cells.
“This is one of the hallmarks of something going from an endosymbiont to an organelle,” Zahr said. “They start getting rid of pieces of DNA, their genome gets smaller and smaller, and they start relying on the mother cell to carry those gene products — or the protein itself — into the cell.”
Cole worked on proteins to study. He compared the proteins found within the isolated UCYN-A with those found in the entire algal host cell. He found that the host cell makes proteins and labels them with a specific amino acid sequence, which tells the cell to send them to the nitroplast. The nitroblast then imports and uses the proteins. Cole identified the function of some proteins, which fill in gaps in specific pathways within UCYN-A.
“It's kind of like a magic jigsaw puzzle that fits together and works together,” Zahr said.
In the same paper, researchers from the University of California, San Francisco showed that UCYN-A replicates in conjunction with the algal cell and is inherited like other organelles.
Change perspectives
These independent lines of evidence leave no doubt that UCYN-A has transcended the role of symbiont. While mitochondria and chloroplasts evolved billions of years ago, nitrogenoplasts appear to have evolved about 100 million years ago, providing scientists with a new, more modern perspective on organelle formation.
The organelle also provides insight into ocean ecosystems. All living organisms need nitrogen in a biologically usable form, and UCYN-A is globally important for its ability to fix nitrogen from the atmosphere. Researchers have found it everywhere from the tropics to the Arctic Ocean, and it fixes a large amount of nitrogen.
“He's not just another player,” Zehr said.
This discovery also has the potential to change agriculture. The ability to make ammonia fertilizers from atmospheric nitrogen allowed agriculture — and the world's population — to take off in the early twentieth century. This process is known as the Haber-Bosch process, and it enables the production of about 50% of the world's food. They also produce huge amounts of carbon dioxide: about 1.4% of global emissions come from this process. For decades, researchers have tried to discover a way to incorporate natural nitrogen fixation into agriculture.
“This system is a new perspective on nitrogen fixation and may provide clues on how to engineer such an organelle into crop plants,” Cole said.
But many questions about UCYN-A and its algal host remain unanswered. The researchers plan to delve deeper into how UCYN-A and algae work and study different strains.
Kendra Turk-Cobo, an assistant professor at UC Santa Cruz, will continue the research in her new lab. Zahr expects scientists will find other organisms with similar evolutionary stories to UCYN-A, but as the first of its kind, this discovery is one for the textbooks.
more information:
Tyler H. Cole et al., A nitrogen-fixing organelle in marine algae, Sciences (2024). doi: 10.1126/science.adk1075
Francisco M. Cornejo-Castillo et al., Metabolic trade-offs constrain cell size ratio in nitrogen-fixing symbiosis, cell (2024). doi: 10.1016/j.cell.2024.02.016

“Web maven. Infuriatingly humble beer geek. Bacon fanatic. Typical creator. Music expert.”





More Stories
Ryan Preece Penalized After Incident With Ty Gibbs at Texas Motor Speedway
Scientists Capture Elusive “Glowing Trees” Phenomenon During Thunderstorms
Satellite Images Reveal Newly Identified Driver of Greenland Glacier Melt